-
E-mail
khjx-hz@163.com
-
Phone
13606716987
-
Address
Jiangdong Industrial Park, Xiaoshan Economic Development Zone, Hangzhou City
Product Categories
Hangzhou Kehao Machinery Co., Ltd
Liquid preparation tank, liquid preparation tank, configuration tank
NegotiableUpdate on 02/11
- Model
- Nature of the Manufacturer
- Producers
- Product Category
- Place of Origin
Overview
The drug preparation tank, preparation tank, and configuration tank produced by Kehao Company are widely used in the preparation, dissolution, heating, cooling, insulation, cultivation, sterilization, and storage of biopharmaceutical production processes
Product Details
Liquid preparation tank/liquid preparation tank/configuration tank
Kehao CompanyproductionLiquid preparation tank/liquid preparation tank/configuration tankWidely used in the formulation, dissolution, heating, cooling, insulation, cultivation, sterilization, storage, etc. of biopharmaceutical production processes
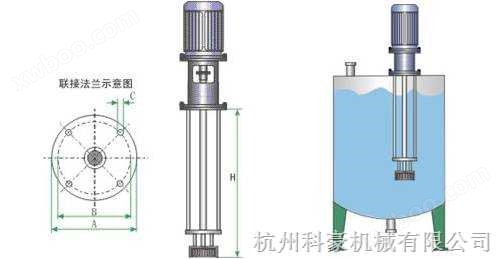
Structure and performance of liquid preparation tank/liquid preparation tank/configuration tank:
This series of liquid tanks strictly comply with the requirements of Article 31 and 32 of the GMP General Principles of the "Good Manufacturing Practice for Drugs" (1998 revised edition), and are designed, manufactured, and accepted in accordance with the requirements of "Steel Pressure Vessels" (GB150-1998) and "Steel Welded Atmospheric Pressure Vessels" (JB/T4735-1997).
● High temperature resistant and anti-aging polyurethane foam or pearl wool shall be used as the container insulation material.
The interface adopts a universal standard quick release chuck type, and the inner liner is made of imported 316L or 304. The inner surface is mirror polished with Ra ≤ 0.28 μ m, and the outer surface is polished with matte, mirror, sandblasted or cold-rolled primary color matte.
The sealing of the mixer adopts imported sanitary mechanical mixing seal or magnetic fully sealed mixing device, and the mixing speed can be selected.
There are liquid level gauges (capacitive, non-contact ultrasonic or glass tube), air breathing ports, thermometers (digital or dial), CIP cleaning ports, sight glasses, explosion-proof lights, SIP sterilization ports, inlet and outlet ports, and manholes at the pipe mouth.
The capacity is available in specifications ranging from 100L to 20000L, and can also be designed and manufactured according to the actual needs of customers.
Randomly attach all GMP validation materials (including material reports, certificates of conformity for purchased parts, validation forms, etc.).
If you need a magnetic stirring tank, please see hereMagnetic stirring mixing tank
If you need a pharmaceutical dispensing tank, please see hereMixing tank, dilution tank, concentration tank
If you need a pharmaceutical dispensing tank, please see hereMixing tank, dilution tank, concentration tank